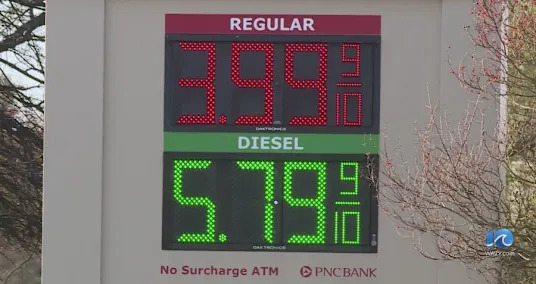(Reuters) -The U.S. Food and Drug Administration is probing the death of a patient who developed harmful antibodies after taking Takeda Pharmaceuticals' blood disorder therapy, the health regulator said on Friday.
The pediatric patient died about 10 months after starting Takeda's drug Adzynma as a preventive therapy, the agency said.
The child had congenital thrombotic thrombocytopenic purpura (cTTP), an inherited condition that causes blood clots in small vessels and can lead to organ damage.
The FDA said the child developed antibodies that blocked the activity of ADAMTS13, an enzyme critical for blood clotting.
Takeda did not immediately respond to Reuters request for comment.
Adzynma, approved in 2023 as the first therapy for cTTP, replaces the ADAMTS13 protein to help prevent dangerous blood clots.
The agency added it has received multiple postmarketing reports of patients developing neutralizing antibodies to ADAMTS13 after treatment with Adzynma.
(Reporting by Kamal Choudhury in Bengaluru; Editing by Vijay Kishore)
latest_posts
- 1
 The most effective method to Look at Medical caretaker Compensations Across Various Clinics
The most effective method to Look at Medical caretaker Compensations Across Various Clinics - 2
 What loving-kindness meditation is and how to practice it in the new year
What loving-kindness meditation is and how to practice it in the new year - 3
 Presenting Nintendo's New Pastel Bliss Con Tones for Switch Gamers: 3 Smart Choices
Presenting Nintendo's New Pastel Bliss Con Tones for Switch Gamers: 3 Smart Choices - 4
 'Stranger Things' series finale trailer shows Hawkins gang gearing up for last battle with Vecna
'Stranger Things' series finale trailer shows Hawkins gang gearing up for last battle with Vecna - 5
 Anthony Joshua's driver charged over Nigeria crash that killed two
Anthony Joshua's driver charged over Nigeria crash that killed two
 NASA's Apollo 8 moonshot saved 1968. Could Artemis 2 do the same in 2026?
NASA's Apollo 8 moonshot saved 1968. Could Artemis 2 do the same in 2026? Countdown to Artemis II: What to know about NASA's moon mission
Countdown to Artemis II: What to know about NASA's moon mission Pain at the pump for Hampton Roads residents
Pain at the pump for Hampton Roads residents These 3 Nail-Free Finds Completely Transformed My Drab Bathroom
These 3 Nail-Free Finds Completely Transformed My Drab Bathroom These men carry towers of birds through Mexico's streets. They say their tradition is dying out.
These men carry towers of birds through Mexico's streets. They say their tradition is dying out. Instructions to Pick the Right Toothbrush for Your Teeth
Instructions to Pick the Right Toothbrush for Your Teeth A Pompeii site reveals the recipe for Roman concrete. It contradicts a famous architect’s writings
A Pompeii site reveals the recipe for Roman concrete. It contradicts a famous architect’s writings 2 ways you can conserve the water used to make your food
2 ways you can conserve the water used to make your food Manual for Individual accounting Rudiments for Fledglings
Manual for Individual accounting Rudiments for Fledglings